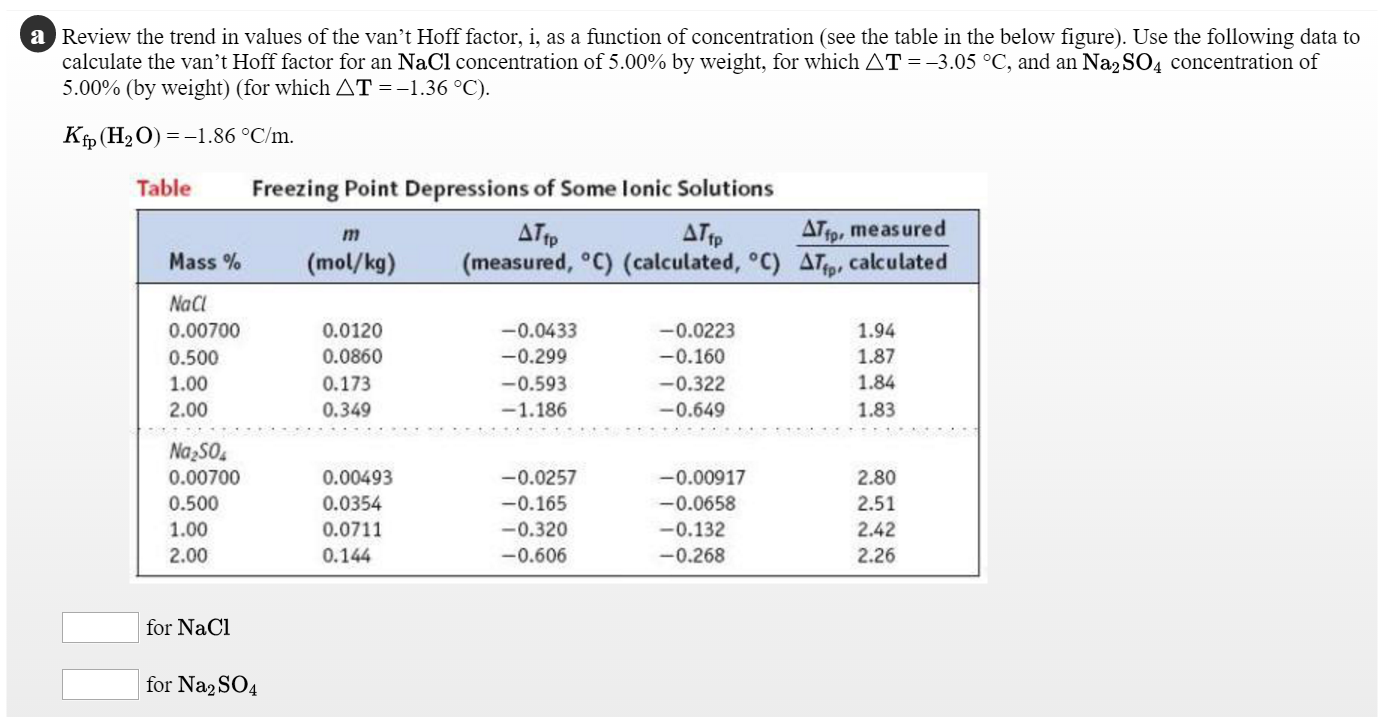
SOLVED: 5. What information does the van't Hoff factor provide? 6. What is the expected van't Hoff factor for each of the following: ZnSO4 NaCl CaCl2

Van't Hoff factor as a function of concentration (calculated according... | Download Scientific Diagram
128. Which one has same Van't Hoff factor i as that of Hg2Cl2: (1) NaCl (3) Al(NO3)3 (2) Na2SO4(4) Al,(SO4)3

Predicting Van't Hoff (i) Factors: Colligative Properties | Chemistry worksheets, Chemistry, Teaching chemistry

SOLVED: 30.0 g of NH4NO3(s) is dissolved in 200 mL of water. If the van't Hoff factor of the resulting solution is 1.9, then what is the freezing point (in °C) of