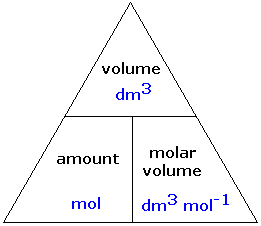
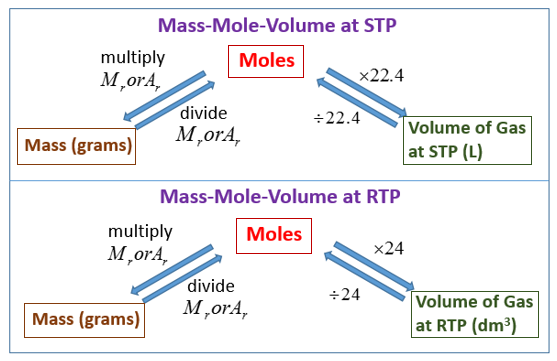
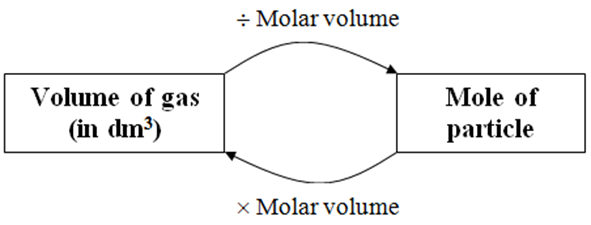
The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download
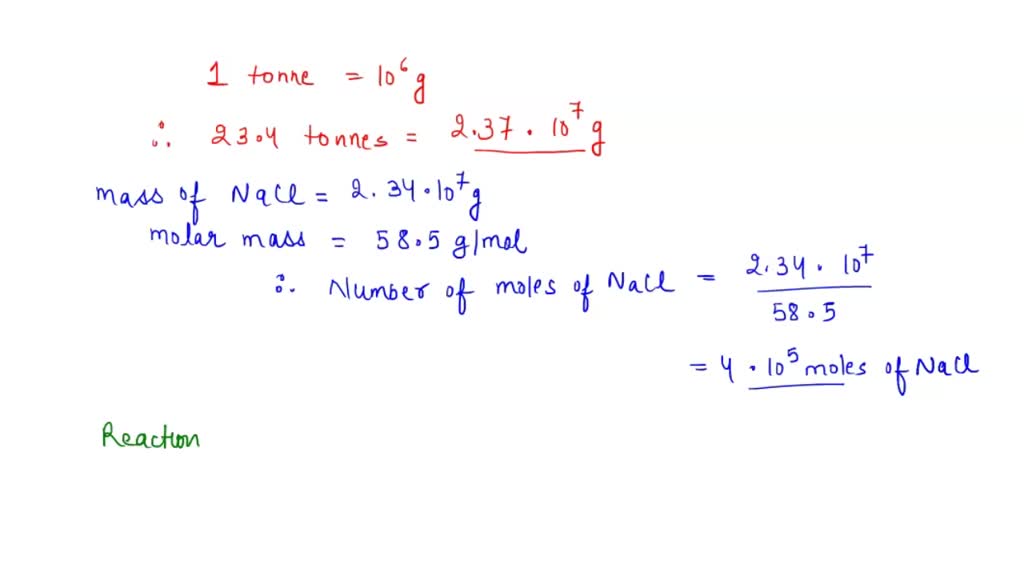
SOLVED: Calculate the maximum volume, in dm³, of chlorine gas at RTP that can be obtained from 23.4 tonnes of molten sodium chloride. (Molar mass of NaCl = 58.5 g/mol) (Molar volume
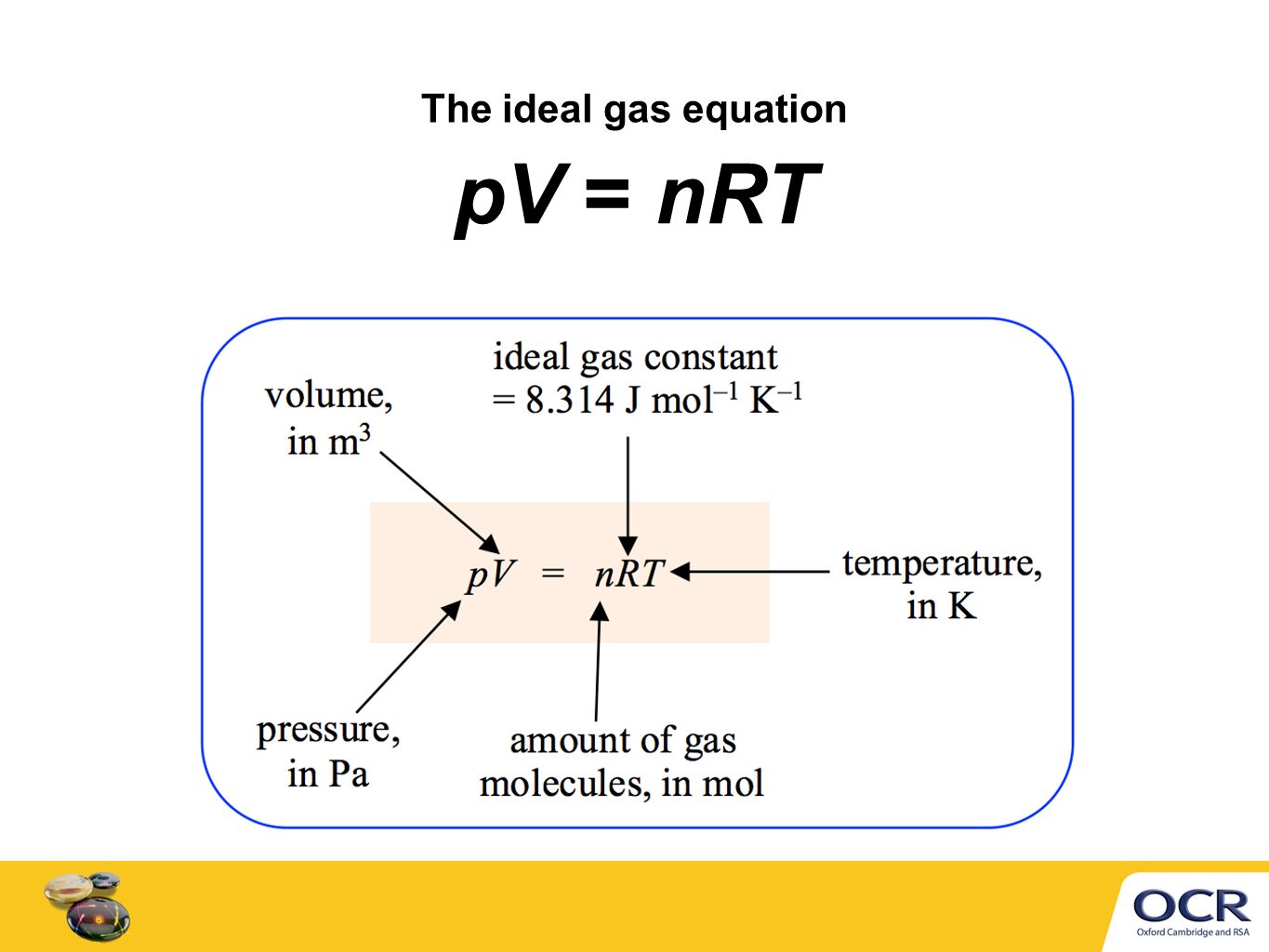
The ideal gas equation. Room temperature and pressure, RTP Limitations At RTP, 1 mol of gas molecules occupies 24.0 dm 3 Conditions are not always room. - ppt download
IGCSE Chemistry 2017: 1.35C: Understand How to Carry Out Calculations Involving Gas Volumes and the Molar Volume of a Gas ( 24 dm3 and 24 000 cm3 at Room Temperature and Pressure (rtp))
![High school chemistry-molar volume] What did I do wrong? The answer is A apparently but I don't get it : r/HomeworkHelp High school chemistry-molar volume] What did I do wrong? The answer is A apparently but I don't get it : r/HomeworkHelp](https://i.redd.it/lrfk0il1k59a1.png)
High school chemistry-molar volume] What did I do wrong? The answer is A apparently but I don't get it : r/HomeworkHelp