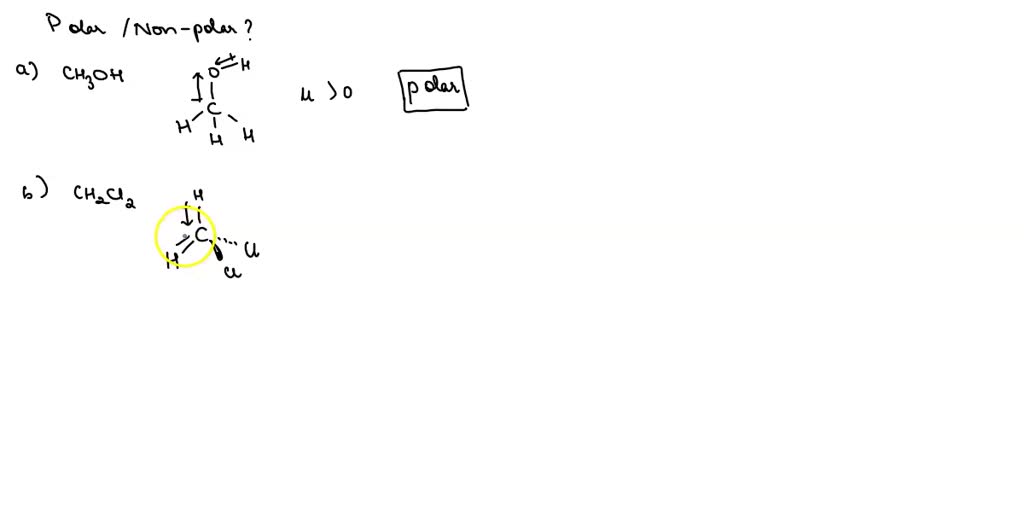
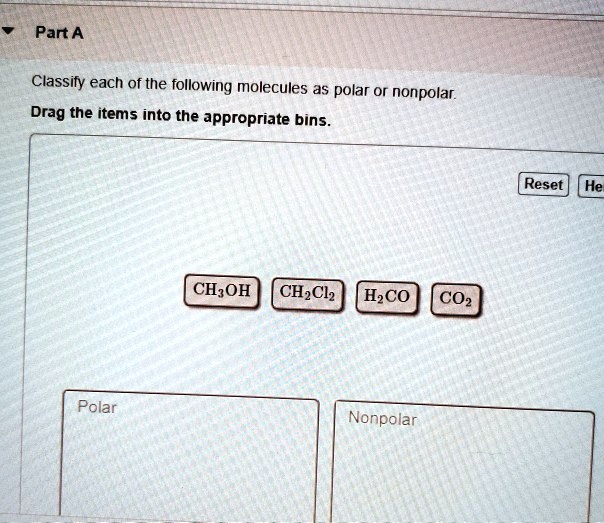
SOLVED: Classify each of the following molecules as polar or nonpolar. Drag the items into the appropriate bins. Reset He CH3OH CH2Cl2 H2CO CO2 Polar Nonpolar
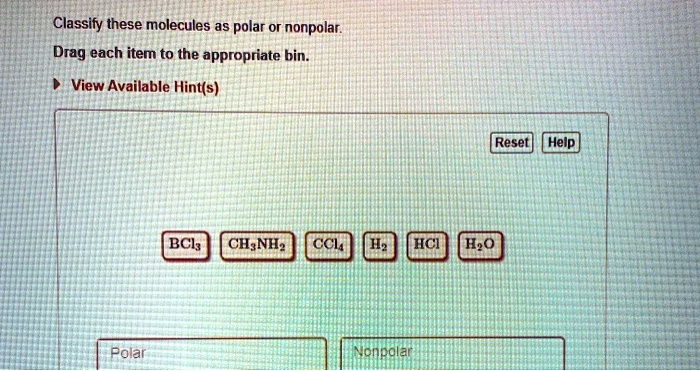
SOLVED: Classify these molecules as polar or nonpolar: Drag each item to the appropriate bin. View Available Hint(s) Reset | Help BCl3 CH4 NH3 CCl4 HCl He Polar Nonpolar
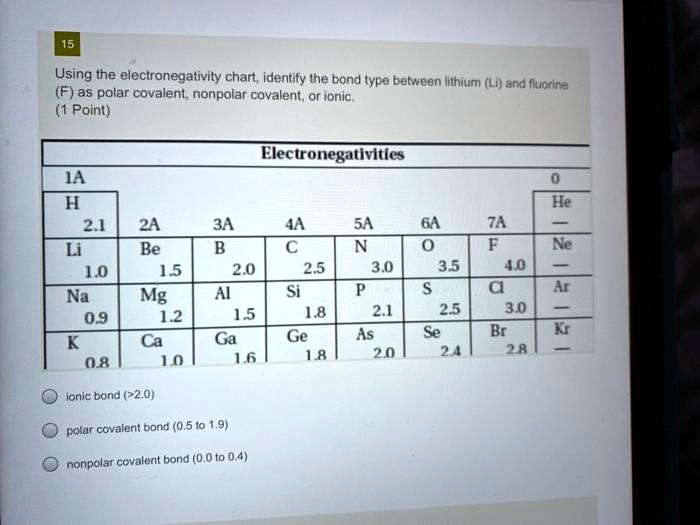
SOLVED: Using the electronegativity chart, identify the bond type between lithium (Li) and fluorine (F) as polar covalent, nonpolar covalent, or ionic. Electronegativities: IA H 2.1 Li 1.0 Na 0.9 K 0.8
What is the difference between a polar covalent bond, a nonpolar covalent bond, and an ionic bond? - Quora
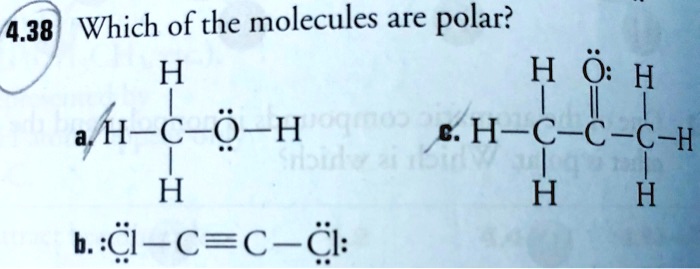
SOLVED: 4.38 Which of the molecules are polar? H H H a H-€- -H E: H-C-C-C-H 1 / e | H H H b..C-C=C-C:

SOLVED: Indicate the type of bonding you would expect between the following elements: Hand F polar covalent Nand F nonpolar covalent He and F ionic Na and F polar covalent nonpolar covalent




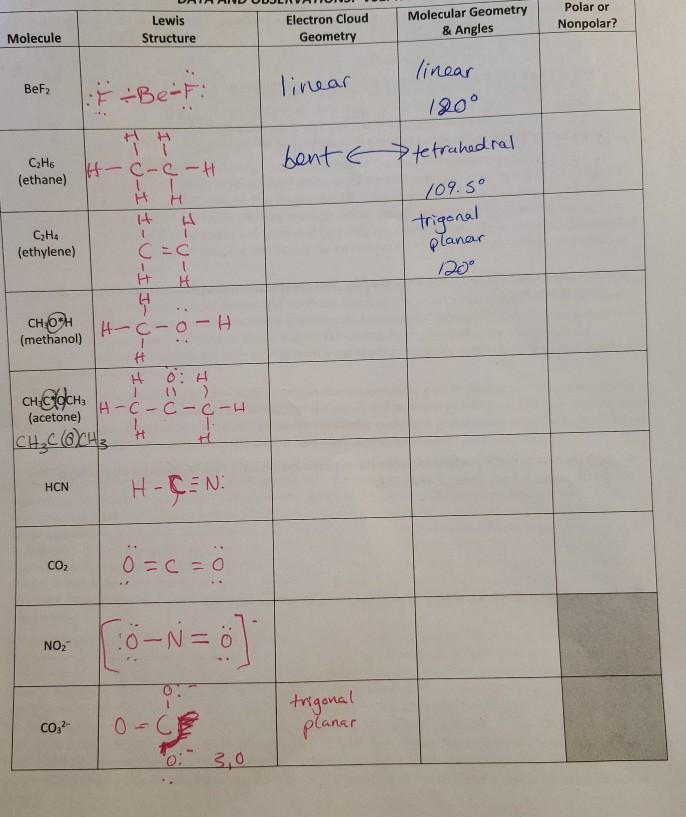
![Solved] Just as a bond can be polar or nonpolar, | SolutionInn Solved] Just as a bond can be polar or nonpolar, | SolutionInn](https://s3.amazonaws.com/si.experts.images/questions/2023/02/63ef5ef5b2554_1676631747640.png)








:max_bytes(150000):strip_icc()/_Benzene-58ee68ef3df78cd3fc238a3b.jpg)