What is the compressibility factor? What is its value an ideal gas? How does it to understand the extent of deviation of a gas from ideal behavior?

Compressibility factor, Z of a gas is given as Z= pVnRT(i) What is the value of Z an ideal gas?(ii) For real gas what will be the effect on value of Z

Aim: What are the properties of Gases?. Compressibility Compressibility is measure of how much volume decreases under increased pressure. Gases are easily. - ppt download
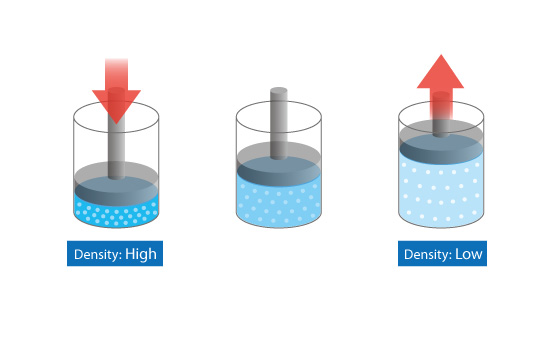
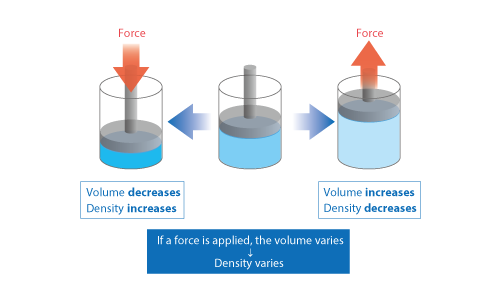
Basic Course of Thermo-Fluid Analysis 06: Chapter 3 Basics of Flow - 3.2.1 Compressible/incompressible fluids|List

SOLVED: Please write the letters, words, and numbers clearly. Compressibility: Define the adiabatic compressibility and isothermal compressibility as: Ks = 1/V KT = VOP|T Ks is the compressibility during an adiabatic process,

SOLVED: Question 3: Compressibility: Define the adiabatic compressibility and isothermal compressibility as β ad and β is, respectively. Show that where Cv and Cp are the heat capacity at constant volume and